Prometheus Bio Inc.
Drug Screen Test Cassette (Saliva) – Multi-Panel Rapid Oral Fluid Drug Detection Kit
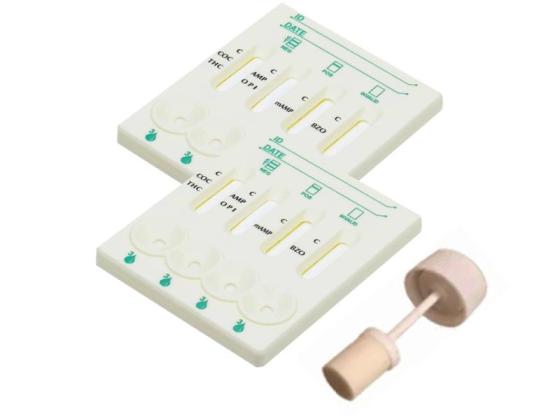
Drug Screen Test Cassette (Saliva) – Multi-Panel Rapid Oral Fluid Drug Detection Kit: Quickly detects multiple drugs from oral fluid samples.
A rapid, screening test for the simultaneous, qualitative detection of Drugs and their metabolites in human oral fluid that can be performed without the use of an instrument. It is an ideal choice for government agencies, law enforcement agencies, and criminal justice departments. The advantages of oral drug testing include low cost, environmental friendliness, non-invasive, and no adulteration.
Features of Drug Screen Test Cassette (saliva cotinine test)
Lower cost, cost-effective, making them an affordable option for drug testing.
Non invasive and dignified collection
Avoid transgender observation
Quick results, read results within 5 minutes
High accuracy: 99% accuracy, comparable to laboratory-based tests.
Avoiding adulteration and pollution to the greatest extent possible
Shelf life: 24 months long shelf life and can be stored easily
Product Specifications
- Product Date 2026
- Delivery cycle within 1 month
- Customize Support
- Price Quotation
- Exit Support
- OEM Quotation
- Supplier Prometheus Bio Inc.
- Delivery Method Land transport/Ocean transport, etc.
- User Manual provide
- Shippable Areas Southeast Asia and Global
Procurement Recommendation
When sourcing multi-panel oral fluid drug test cassettes, prioritize suppliers that comply with international regulatory standards such as CE marking under EU IVDR, FDA 510(k) clearance (if applicable), or ISO 13485 certification for medical device quality management. The kit should demonstrate high sensitivity and specificity across all targeted analytes—commonly including THC, cocaine, opioids, amphetamines, and benzodiazepines—with clearly documented cross-reactivity data. Ensure the supplier provides batch-specific validation reports, stability studies (typically 12–24 months at room temperature), and instructions validated for non-clinical settings like workplace screening, law enforcement, or rehabilitation programs. Packaging must be tamper-evident, individually sealed, and include clear procedural graphics to minimize user error. Consider ease of sample collection: integrated oral swabs should be comfortable, yield sufficient fluid volume (usually ≥100 μL), and feature visual saturation indicators. Shelf life, storage conditions (avoiding extreme humidity or heat), and lot traceability are critical for audit readiness. Avoid kits making unsubstantiated “instant lab-equivalent” claims without third-party verification. For forensic or legal defensibility, confirm whether the product supports confirmatory testing pathways (e.g., GC-MS referral). Finally, evaluate supplier responsiveness in providing technical documentation, training materials, and post-market support, especially when deploying tests across multiple jurisdictions with varying drug screening regulations. (for reference only)
Due to market conditions, product updates, and policy changes, please contact the supplier for the latest information.








Comments